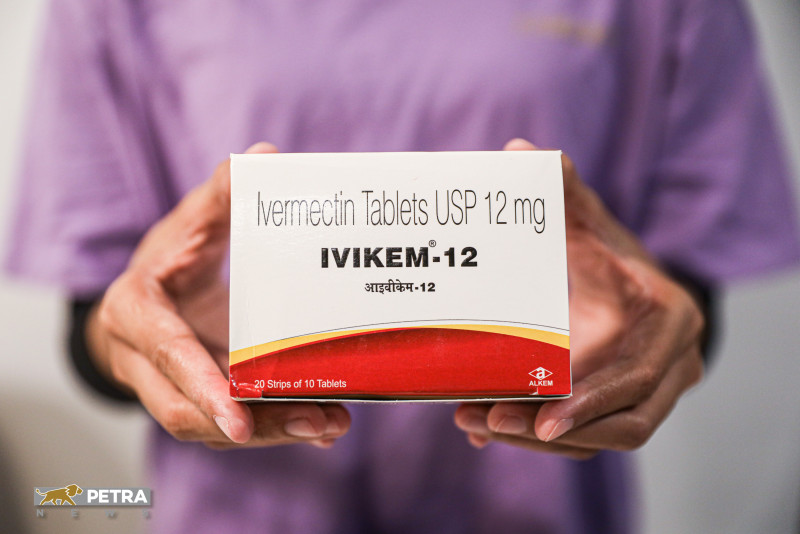
KUALA LUMPUR – The usage of ivermectin as a form of treatment during early stages of Covid-19 was found to not prevent patients from progressing to more severe phases of the disease, according to a recently published study.
Research conducted by Malaysia’s Ivermectin Treatment Efficacy in Covid-19 High-risk Patients (I-Tech) study group concluded that, in its randomised clinical trial of high-risk patients with mild to moderate Covid-19, no evidence was found to reinforce the effectiveness of ivermectin treatment.
Carried out at 20 government hospitals and a Covid-19 quarantine centre between May 31 and October 25 last year, the study was approved by the Medical Research and Ethics Committee and saw the enrolment of 500 patients.
None of the 13 deaths of patients in the trial were attributed to treatment with the anti-parasitic veterinary drug, with severe Covid-19 pneumonia being the principal direct cause for nine deaths and four from nosocomial sepsis.
Thirty-three patients from the ivermectin group, however, reported adverse reactions to the drug, with diarrhoea being the most common effect. Consequently, six patients discontinued the usage of ivermectin and three withdrew from the study.
“The study findings do not support the use of ivermectin for patients with Covid-19.
“Prior randomised clinical trials of ivermectin treatment for patients with Covid-19 and with 400 or more patients enrolled focused on outpatients.
“In contrast, the patients in our trial were hospitalised, which permitted the observed administration of ivermectin with a high adherence rate,” said the study that was published yesterday.
The study had enrolled confirmed Covid-19 positive patients who were 50 years old or older with at least one comorbidity and in categories 2 or 3 of the Covid-19 severity stage.
The ivermectin dosage for each patient in the intervention arm was calculated to the nearest 6mg or 12mg whole tablets. The first dose of ivermectin was administered after randomisation on day one of enrolment, followed by four doses on days two through five.
The results of the study align with what medical professionals and bodies have been saying so far on there being no evidence to support the use of ivermectin to treat Covid-19.
The study was published in Jama Internal Medicine, a peer-reviewed medical journal.
The European Medicines Agency and the US Food and Drug Administration said data showing the effectiveness of ivermectin to treat Covid-19 is lacking, and that the drug is not authorised for use in treating Covid-19 outside clinical trials.
The proposed usage of ivermectin came under the spotlight in April 2020, after researchers from Australia’s Monash University found that it prevented the Covid-19 virus from replicating in cell culture within two days.
Last July 28, the Malaysian Pharmacists’ Society and Malaysian Medical Association urged the authorities to crack down on the widespread illegal sale of ivermectin nationwide, saying the public must understand that its uncontrolled use can cause possibly damaging side effects.
On the other hand, the Consumers’ Association of Penang said it supports the Malaysian Alliance for Effective Covid-19 Control, which has been lobbying for the use of ivermectin under emergency use authorisation alongside the country’s current immunisation programme. – The Vibes, February 19, 2022