KUALA LUMPUR – The Drug Control Authority has approved the Johnson & Johnson and CanSino Covid-19 vaccines for emergency use, said Health Director-General Tan Sri Dr Noor Hisham Abdullah.
The agency also gave the nod for the Pfizer-BioNTech jab to be used on children aged 12 and above under the National Covid-19 Immunisation Programme.
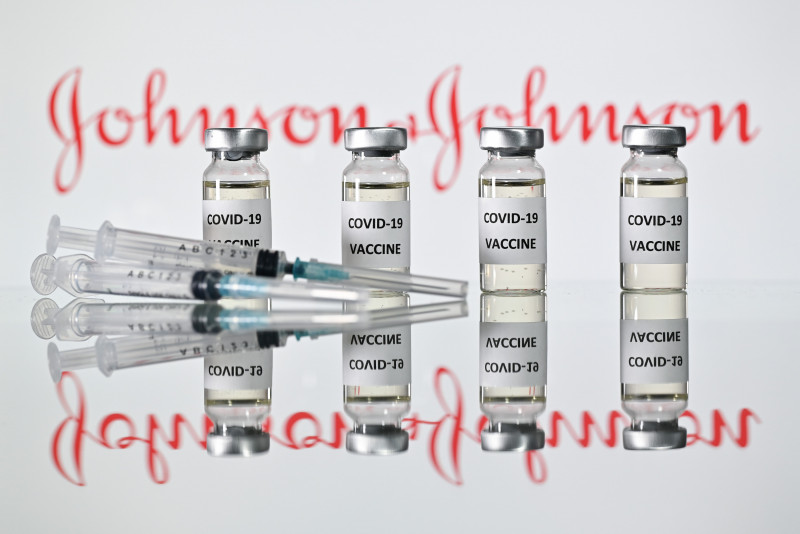
In a statement today, Dr Noor Hisham said the Johnson & Johnson vaccine, called Janssen, will be procured via the Covax facility headed by the World Health Organisation (WHO).
Both the Johnson & Johnson and CanSino vaccines are single-dose jabs and have received Emergency Use Listing approval from WHO.
The Pfizer-BioNTech, Sinovac and AstraZeneca jabs currently used in Malaysia are a two-dose regimen.
“Despite the approval given to the Pfizer-BioNTech vaccine for use on those aged 12 and above, the Health Ministry will still prioritise high-risk groups for vaccination under existing national immunisation programme policies,” said Dr Noor Hisham.
“The approval of this conditional nod requires data on quality, safety and effectiveness to be evaluated and monitored over time. It is to ensure a positive benefit over risks for the vaccines.” – The Vibes, June 15, 2021



.jpg)
.jpg)