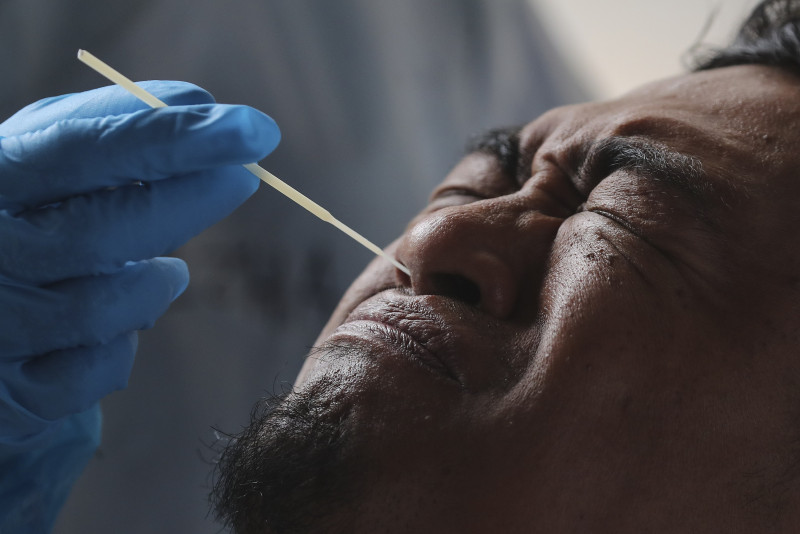
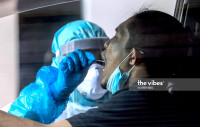
KUALA LUMPUR – The Malaysian Medical Device Authority has given a conditional approval for the import and distribution of the Covid-19 self-test kits for public use, said Health Director-General Tan Sri Dr Noor Hisham Abdullah.
In a statement today, he said this will alleviate the burden on frontliners while easing the detection and control of Covid-19 cases.
Among the regulations imposed on self-test kit importers and distributors are to provide clear and user-friendly instructions for usage and disposal methods, implement a test kit safety and performance monitoring system, and test kit distribution records.
So far, two test kits have passed the technical and testing procedures and will be given approval for the import and distribution nod in the near future.
Meanwhile, another 15 kits are undergoing performance testing.
“Seeing that the test kits are for public use, these conditions are vital to provide a control mechanism to ensure their safety and performance.
“The results derived from these kits can be used by the public as an immediate way to find out their Covid-19 status. However, this kit cannot be used to replace the RT-PCR test as a diagnostic tool.”
The self-test kits that have been given conditional approval will be displayed on the MDA website and the list will be updated from time to time for public reference.
Companies that wish to apply for the conditional approval must have establishment licences issued under the MDA Act 2012 (Act 737) while the kits they sell must go through technical evaluation and performance testing. – The Vibes, July 14, 2021







_and_lord_mayor_rajendran_posing_in_a_mock_up_lrt-Ian_pic.jpeg)





.jpeg)











