KUALA LUMPUR – The Health Ministry has issued an immediate recall on 14 products containing the opioid pholcodine that are registered with the Drug Control Authority (DCA) following reports of adverse effects.
The DCA is also cancelling the registration of the products in the recall exercise and ordered for them to be disposed of by June 20, according to a document sighted by The Vibes.
Affected product-registration holders can appeal the directive by April 3.
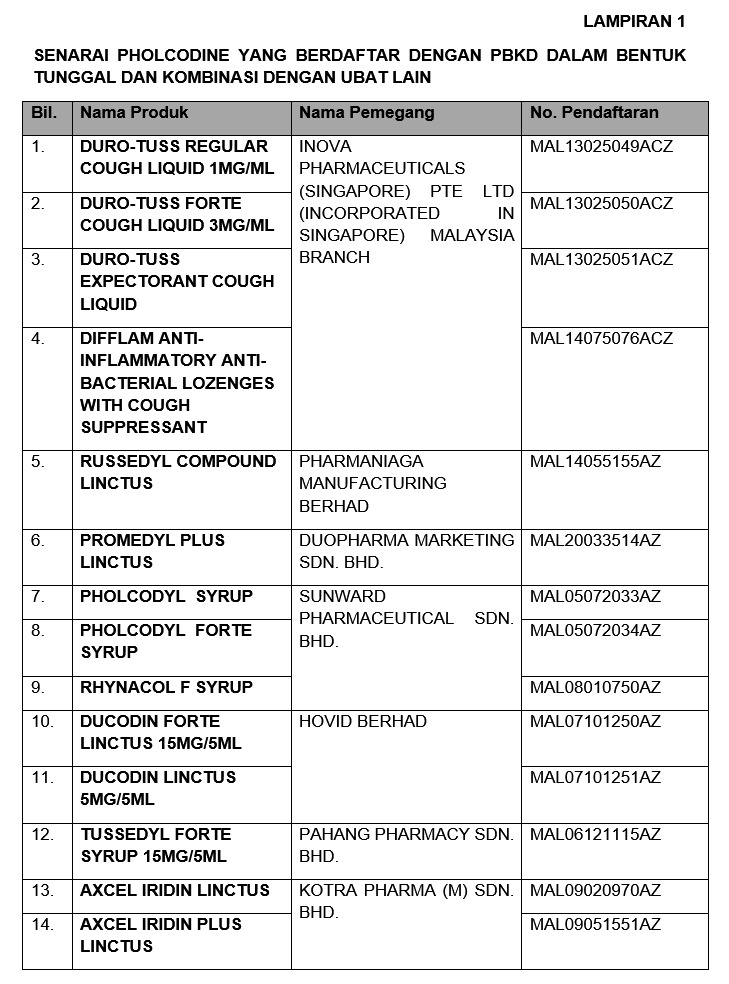
Among the affected products are three types of Duro-Tuss cough liquids, Difflam anti-inflammatory anti-bacterial lozenges with cough suppressant, Pholcodyl syrup, Tussedyl Forte Syrup, and other cough medications.
In a statement, Health Director-General Tan Sri Dr Noor Hisham Abdullah said all affected product-registration holders are responsible for informing pharmacies and clinics to immediately halt sales of the affected products and return them to suppliers.
He added that medical officers are to immediately stop dispensing, selling, and distributing all medication containing pholcodine.
Those who had taken medication containing pholcodine in the past 12 months are advised to inform their doctors prior to surgeries, especially involving general anaesthesia.
Meanwhile, he advised patients with non-productive cough to seek alternative medications, such as those containing dextromethorphan.
There are more than 40 dextromethorphan products registered with the DCA.
The National Pharmaceutical Regulatory Agency received 12 reports on 17 adverse effects after the use of pholcodine while there are no reports of anaphylactic reaction.
Noor Hisham said it was found that several medications containing pholcodine – usually to treat “non-productive/dry, irritating” coughs in adults and children – pose safety risks.
“This means, those who have taken medication containing pholcodine (typically cough medication) in the past 12 months are at a higher risk of anaphylaxis if they are given muscle relaxants or neuromuscular blocking agents (NMBAs) during general anaesthesia – for example, when they are undergoing an operation.
“The decision by the DCA is based on evidence from the Allergy to Neuromuscular Blocking Agents and Pholcodine Exposure study, post-marketing safety data, and information obtained from medical practitioners, national agencies of other countries such as the UK, Australia, (and) EU countries such as France and Norway where they have recalled products containing pholcodine.”
The World Health Organisation reported a total of 852 reports of adverse effects involving the use of pholcodine. Of those, there were 42 reports of anaphylactic reaction and 20 reports of anaphylactic shock. Of those cases, nine involved the co-use of NMBA (suxamethonium). – The Vibes, March 23, 2023



![[UPDATED] MoH recalls 14 products containing opioid pholcodine](https://media.thevibes.com/images/uploads/covers/_large/13092022_-_KUL_-Health_Ministry_Kementerian_Kesihatan_medicine_medical_policy_generic-_SYEDAIMRAN.JPG)