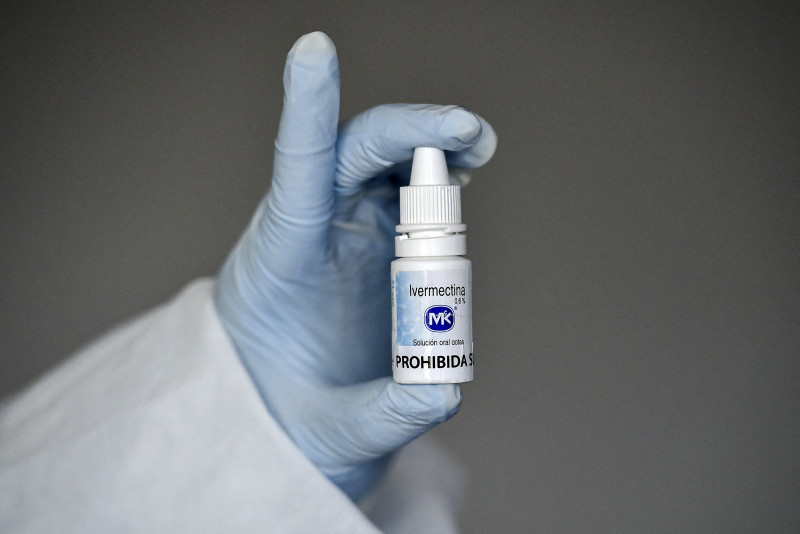
KUALA LUMPUR – The debate on the use of ivermectin as a prophylaxis and treatment for Covid-19 has raged further, with the Malaysian Society of Infection Control and Infectious Diseases (MyICID) expressing concern about the anti-parasitic drug being used for the purpose without clinical endorsement.
In a statement today, it said the drug should not be administered to the public in the fight against the coronavirus before it has been endorsed in clinical guidelines, whether locally or internationally, for the said purpose.
Laboratory evidence, anecdotal reports and early clinical trials have shown ivermectin to demonstrate signs that it could be a potential repurposed drug for Covid-19 prevention and treatment, it said.
We disagree with the premature sentiment that ivermectin is a ‘miracle drug’ for Covid-19 at this juncture, as the evidence that is available is controversial.
“Ivermectin is an anti-parasitic drug with a good efficacy and safety profile. Based on laboratory evidence, anecdotal reports and several early clinical trials, it appears to be a potential repurposed drug for Covid-19 prevention and treatment.
“However, results from well-designed, large, randomised controlled trials are needed to determine the efficacy and safety of Iverivermectinmectin for use against Covid-19.”
On Monday, a group of six medical and consumers’ non-governmental organisations urged the government to immediately approve the use of ivermectin on Covid-19 patients in Malaysia.
In an open letter, 10 medical experts said there is nothing to lose in deploying the tried-and-tested medicine, which is being increasingly used in other countries against new Covid-19 variants.
Ivermectin has been proven safe many times over, with credible studies worldwide confirming that it can reduce the risk of Covid-19 infection by 88% and the mortality rate by 83%, they said.
MyICID pointed out, however, that health authorities, such as the World Health Organisation, United States Food and Drug Administration, Infectious Diseases Society of America, European Medicines Agency and Malaysia’s own Health Ministry have recommended the use of ivermectin only in clinical trials.
“MyICID concurs with this recommendation as this will ensure the safety of patients, while evaluating the effectiveness of ivermectin against Covid-19. To date, there are more than 80 randomised controlled trials on ivermectin that are either completed or ongoing around the world.”
Malaysia is conducting a clinical trial involving 500 high-risk Covid-19 patients.
“The results of these trials will yield robust scientific evidence, not only on the role of ivermectin in Covid-19 treatment, but also on the optimal dose and duration needed for effective outcomes without causing adverse effects to patients,” said MyICID.
It warned against sentiments that could lead to the misuse of and illegal profiteering from the drug.
There have been reports of people acquiring ivermectin from illegitimate sources, or consuming ivermectin meant for animal use, leading to the development of adverse effects.”
It is important for consumers to first understand the risks and benefits of any medication they intend to take, added the group.
“We strongly urge the Malaysian public to heed the advice of and adhere to the treatment recommendations by the Health Ministry and international health agencies, which are rigorously evaluating the efficacy and safety of ivermectin in Covid-19 treatment.” – The Vibes, June 25, 2021